Our Regulatory Affairs Specialists are highly skilled professionals renowned for their exceptional expertise in navigating the complexities of regulatory requirements and delivering comprehensive regulatory support services.
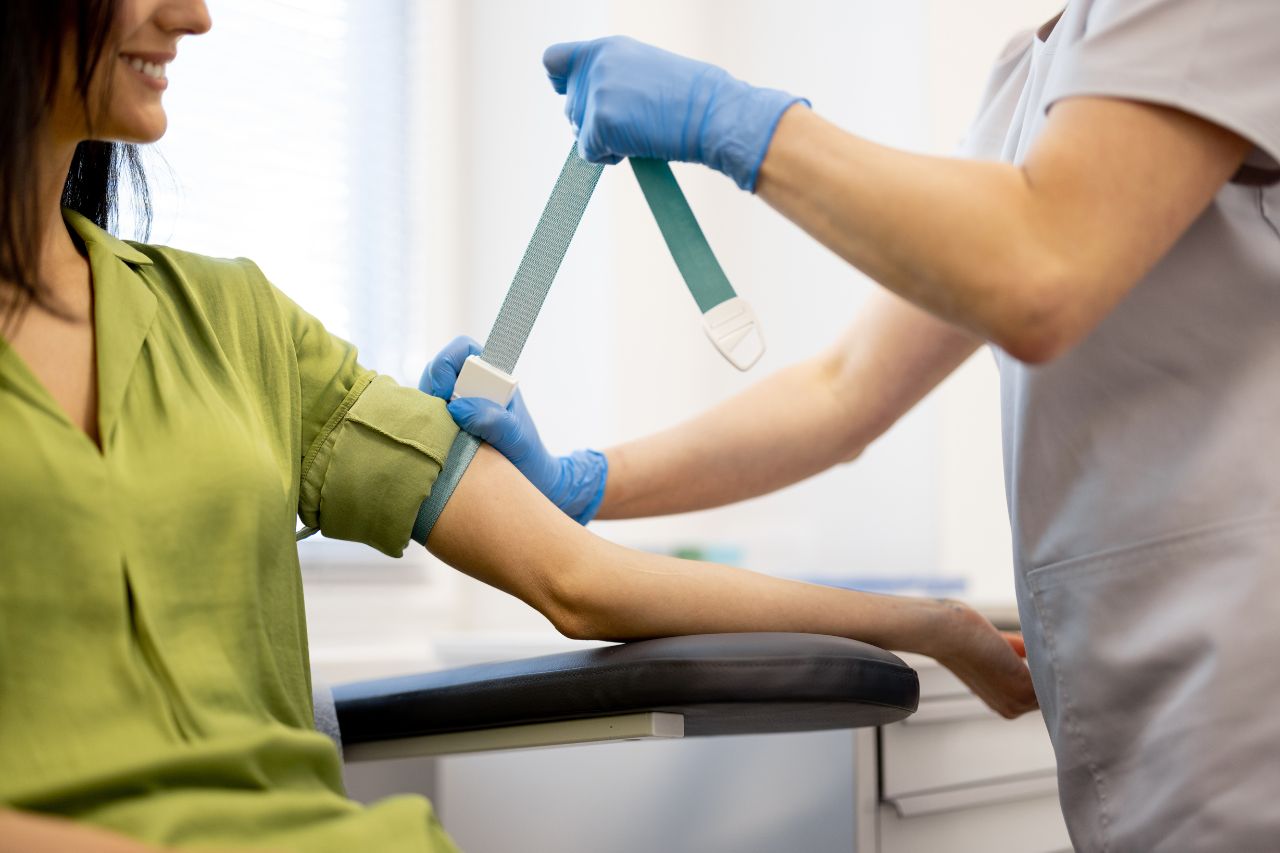
Ensuring regulatory compliance is critical to the success of clinical research. Our robust regulatory affairs services are designed to ensure that clinical trials meet all regulatory requirements and adhere to global standards.
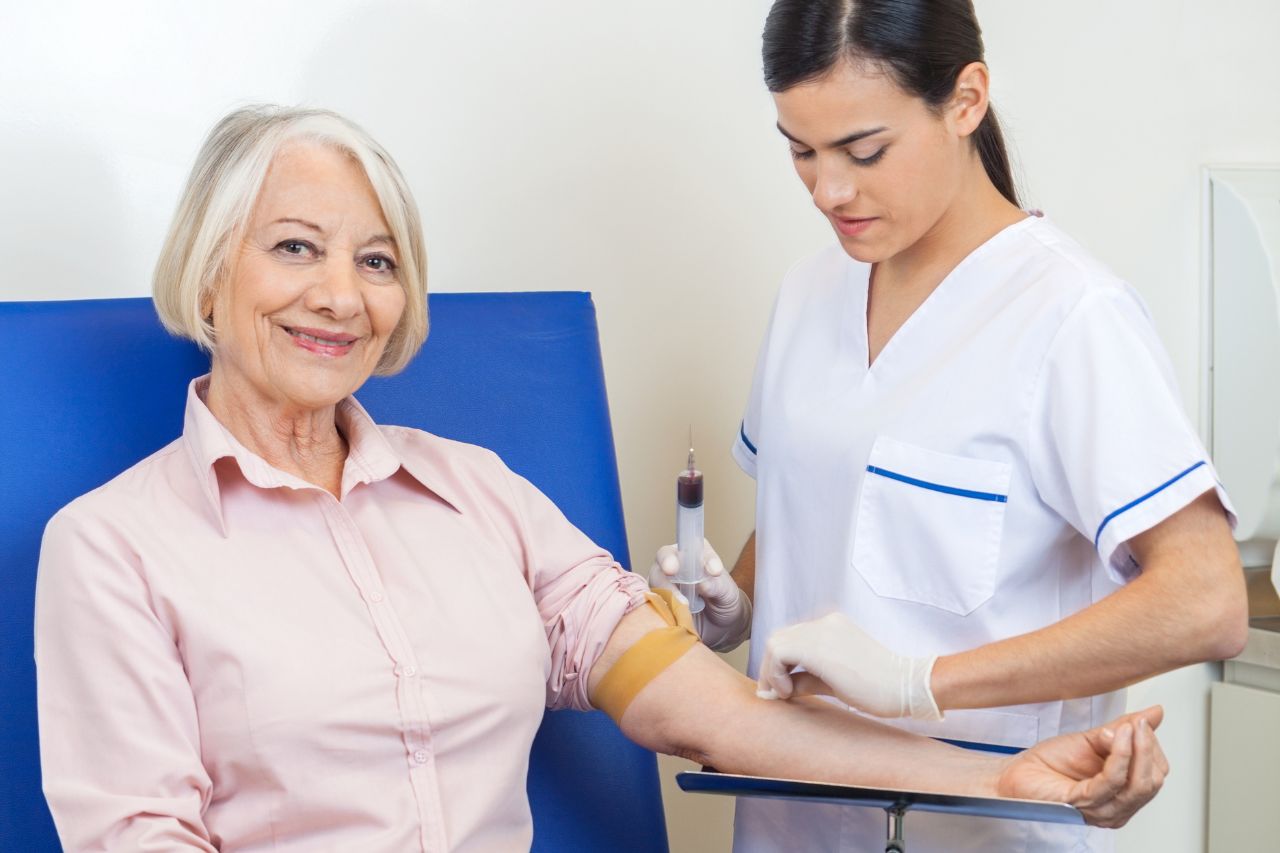
Milagro Clinical Research (MCR) ensures rigorous and ethical regulatory compliance. Our dedicated team meticulously oversees every phase of the trial, guaranteeing adherence to regulatory standards and protocols. We ensure timely submissions, patient safety, and high-quality results through comprehensive and precise regulatory activities.
At Milagro Clinical Research, we are dedicated to unlocking the miracle of medical advancements, surpassing limits to healing.
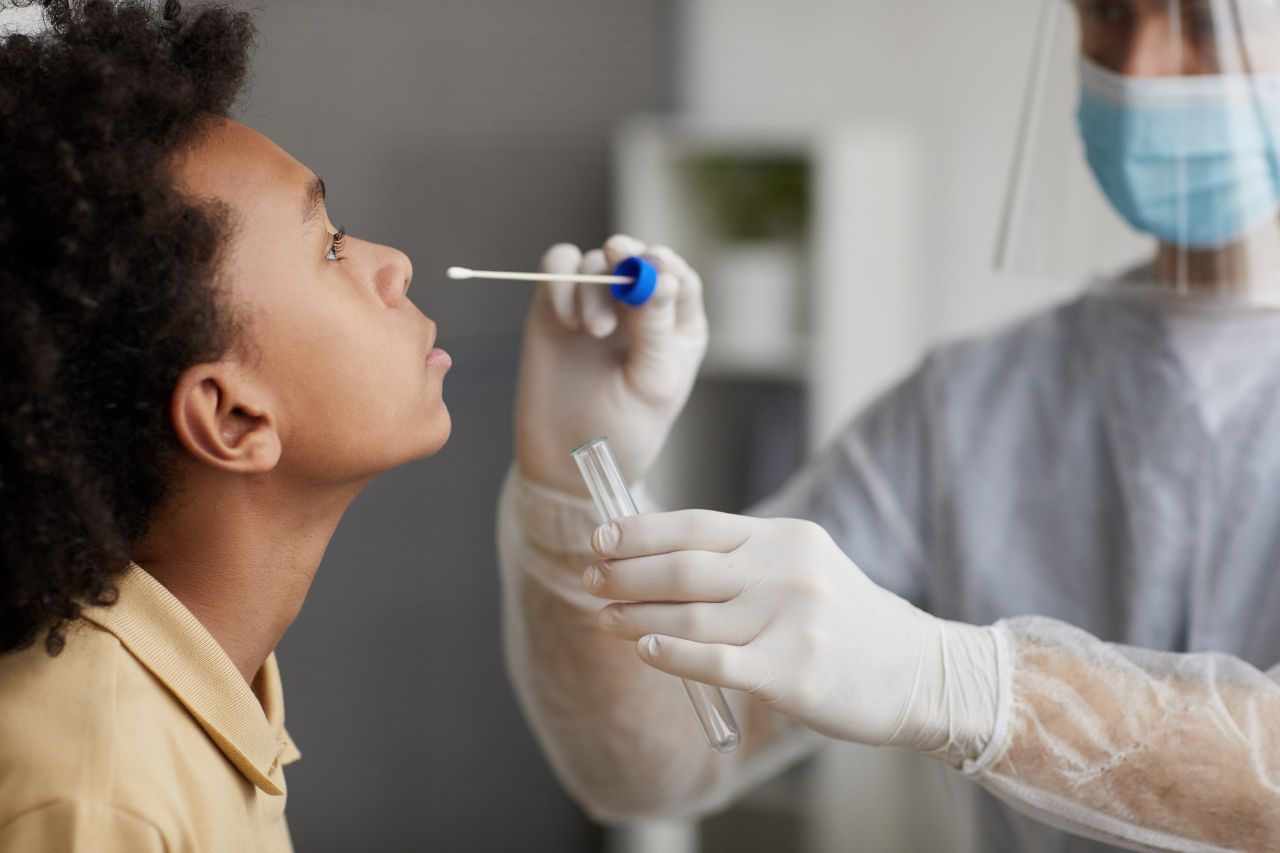
With over two decades of experience across diverse therapeutic areas, our team brings unparalleled expertise to every clinical trial.
We maintain rigorous adherence to global regulatory standards, ensuring every trial meets the highest certifications and ethical standards.
Our state-of-the-art facilities provide the optimal environment for conducting cutting-edge research and delivering precise results.
Supported by expert professionals, we ensure meticulous management of medications and protocols throughout every trial phase.


Office No 306, NBC Complex, Opposite to Sakal Bhavan, Sector 11, CBD Belapur, Navi Mumbai, 400614, Maharashtra
Mail : info@milagrocro.com
Call : +91 9004526456